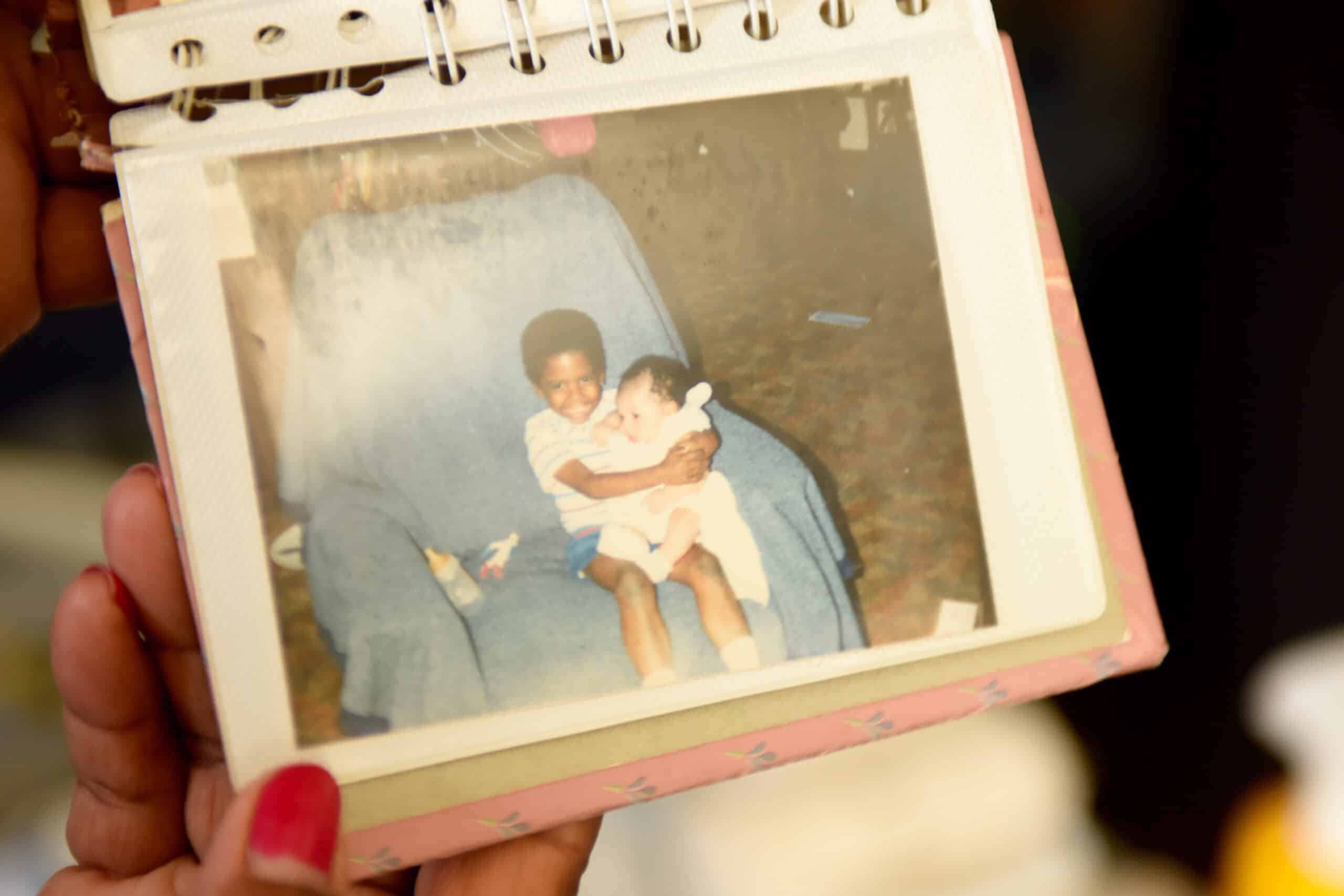
Young Marqus holding Ashley
In 2016, Marqus and I found an NIH study for sickle cell disease and leg ulcers. We came across the study through my carpool friend who worked at the National Institutes of Health (NIH). They remembered that I had a brother with sickle cell and brought me information as we exited the metro.
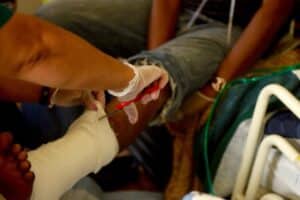
Francesca, Marqus’ mother, caring for his leg ulcer
I called Marqus to explain the protocol and he jumped at the opportunity. By this point, Marqus’ leg ulcers had been open for three years. He was getting them debrided frequently, skin grafts, combination creams, and repeat infections. The open skin got so bad at some points that it was nearly to the bone. The pain was so intense that it would prevent him from walking. We would try anything to contribute to science to not only help Marqus, but to also prevent others from ever having to go through this type of pain.
Part of the study protocol included a wash-out period. That meant that Marqus needed to go off his medications to manage the leg ulcers so that the sample could be fresh. In the end, that led to Marqus’ developing an infection and being hospitalized. He could not enroll in the study. What upset us both the most was that he wouldn’t be included in science nor could we try a new form of treatment that could potentially help the leg ulcers. He recovered though and we continued to look for more trials and studies.

Marqus’ first dose of a new medication
New Treatments Provided Hope: With every new therapy that was approved, Marqus was first in line to access the treatment. We would track the trials via conferences, webinars, and engaging with the companies. When he got his first prescriptions, he wouldn’t fill them immediately. He followed our at-home protocol, of course created by our nurse mom. The steps went like this:
- Read entire packet insert online to understand the risk and benefits
- Start new drug on a Tuesday, during work hours, never after work or on weekends, just in case we had to call his provider
- Fill prescription
- Take first dose when someone else was home
When the new sickle cell treatments got approved – 2017 and 2019 – Marqus even framed the prescriptions to remember the moment. In his lifetime, he always wanted sickle-cell specific treatments to be developed. To our family, new treatments, clinical trials, innovation, however you want to call it, were all steps in the solutions, mitigating the complications of sickle cell disease and ensuring a better future for the next generation. For most of our lives, there hadn’t been any investment or progress focused on treating sickle cell disease, rather than just managing the complications.

Marqus and Ashley traveling from FDA
MY HERO
Marqus passed away June 22, 2020, in the height of the COVID-19 pandemic. His funeral was restricted to just Illinois residents and had to abide by a laundry list of rules outlined to slow the spread of the dangerous virus. Even in his passing, he still signed up to donate his organs to donor banks and committed himself to science. He awed me even in that moment.
A Glimmer of Hope: During the first pandemic winter, I sat at my parents’ home most days and watched the death toll on the side of the screen increase. I watch the virus run rampant through the sickle cell community, getting calls weekly about another sickle cell warrior, family member, family friend, or colleague that were taken by the virus.
Then in 2021 clinical trials started for covid vaccinations. Those vaccinations offered a glimmer of hope that we could get out of the pandemic and save lives. I thought, what would Marqus do? He would be part of the solution. He would be part of innovation. I enrolled into the Novavax PREVENT-19 trial.
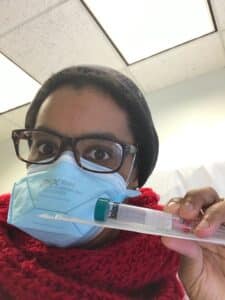
Ashley taking a covid test during clinical trial
My First Clinical Trial: To find the trial, I searched clinicaltrials.gov. I saw openings for two Novavax trial sites in and around Chicago. I called the first site to inquire about enrollment. I chose that site because it was suburban, had a parking lot, and was more isolated from people (think COVID times…Ahh! People!) The study coordinator told me I wasn’t qualified, which I knew to be untrue. I knew that the COVID trials were enrolling participants that matched the United States population and that generally finding trials creates barriers to minority populations.
After getting denied from the first trial site, I called the second trial site, which became my second home. The trial coordinator ran me through eligibility and enrolled me into the trial. They sent me my enrollment package which I reviewed with a few nurse friends who worked on clinical trials. The entire enrollment screening took two days and then I had my first appointment.
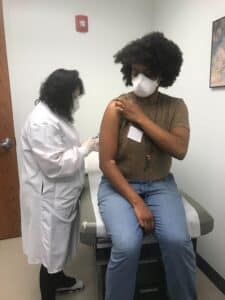
Ashley participating in clinical trial
Trial Day: The trial was a double blind, placebo trial and eventually had a crossover study. That meant, the first two doses of vaccination had a cohort that got saline instead of the drug. After the initial data was collected, due to the severity of COVID-19 and need for vaccination, all participants received another two doses. The participants who received the study drug got saline and the participants who received saline got the study drug.
The visits took 3 hours each. I was always impressed at the number of people who looked like me at the study. It debunked any and every myth that minorities do not enroll in clinical trials. We were all there. Some days I asked other participants why they enrolled. One participant told me that they wanted extra income and were out of work. Another participant told me that they were curious about trial participation. A third participant told me that they wanted to hurry up and get a vaccine and this trial was the fastest way to do that.
I had a total of 5 visits and virtual follow up for a year. During each visit, they would do the following:
- Review the study protocol
- Review of our e-diaries
- Conduct a doctor’s appointment
- Test us for covid
- Test women for pregnancy
- Draw many tubes of blood
- Issue study drug

Quote on wall at clinical trial site ‘Everyday, people from all over the world participate in clinical research trials to help themselves and others improve the quality of life.’
After we received the study drug, we had to wait a half hour to ensure we did not have a severe reaction. I got excited for each appointment. I felt like I was being part of the solution. I was actively doing something to make others’ lives better. I also was protecting my family, protecting myself, and honoring my brother’s commitment to innovation.
A Family Affair: That feeling was not mutual however. My parents were nervous. Given that we had just lost Marqus less than a year prior and the ongoing pandemic, my participation in a clinical trial was nerve wracking. Regardless, nerves aside, my parents understood my desire to participate.
I suspect that I received the study drug for the first two shots because I had side effects. One side effect was extreme fatigue and a fever. Whenever I awoke from sleep, I found a parent hovering over to check that I was breathing. This was a reminder to me that clinical trials and the decision to enroll in them is a family affair. My dad and sometimes both parents drove me to the appointments. They waited in the car to avoid entering the facility. They cared for me during the side effects and even reminded me of side effects when it came time to complete my e-diary. Together, we got me through that trial.

Marqus and Ashley, 2019 Red Cross Blood Service Heroes
In the end, the Novavax vaccine was approved under emergency authorization in 2021 and received full approval in 2023. Since then, I have enrolled in natural history studies. I donate blood and help others understand the clinical trial process outside of my work at Sick Cells. As Marqus said, innovation is the future. We all must be part of the solution.
To learn more about clinical trials, join Sick Cells on November 13, at the Sick Cells Therapeutic Conference, hosted in Washington, DC.
Published Oct 2025

